|
It is the insoluble salt formed as a product of the precipitation reaction. The chemical reaction between potassium chloride (KCl) and silver nitrate (AgNO 3 ), in which solid silver chloride is precipitated out of the solution, is one of the most common examples of precipitation. When two or more two solutions containing different salts are combined, insoluble salts are produced that precipitate out of the solution. Most precipitation reactions are single replacement reactions or double replacement reactions. (Public Domain ZabMilenko) The use of solubility rules require an understanding of the way that ions react. Precipitation reactions are an example of double displacement reactions in which a solid form residue called the precipitate is left behind. Precipitate: In chemistry, a solid formed by a change in a solution, often due to a chemical reaction or change in temperature that decreases solubility of. 1: Above is a diagram of the formation of a precipitate in solution. Reaction AgNO 3(aq) + KCl (aq) AgCl + KNO 3(aq) Redox Reaction Those chemical reactions in which oxidation and reduction take place simultaneously are called redox reactions. The insoluble salts that are formed in precipitation reactions are called precipitates. In precipitation reactions, two soluble salts in aqueous solutions are combined and form an insoluble precipitate. Precipitation Reaction – Definition and MeaningĪ chemical reaction that takes place in an aqueous solution where two ionic bonds combine and an insoluble salt is precipitated out as a byproduct is called the precipitation reaction. Many reactions of this type involve the exchange of ions between ionic compounds in aqueous solution and are sometimes referred to as double displacement, double replacement, or metathesis. Examples of precipitation reactions: Ferrous sulphate and Sodium hydroxide. A precipitation reaction is one in which dissolved substances react to form one (or more) solid products. Therefore, it is referred to as the 'precipitate.’ Precipitation Reactions and Solubility Rules. One formed product is insoluble in the solution and is thus precipitated out. Keeping this in mind, let us talk about Precipitation reactions in these reactions, by combining two different soluble salts in an aqueous state, two different products were formed. Some of the most common examples of chemical reactions are burning, corrosion, cooking of food and digestion etc. We see many chemical reactions in our day to day lives. 
These reactants can be in any state - solid, gaseous, or liquid. Various compounds during the chemical reaction will interact with each other and are called reactants. Interaction of reactants and the products that are involved in a chemical reaction is displayed in the chemical equations. 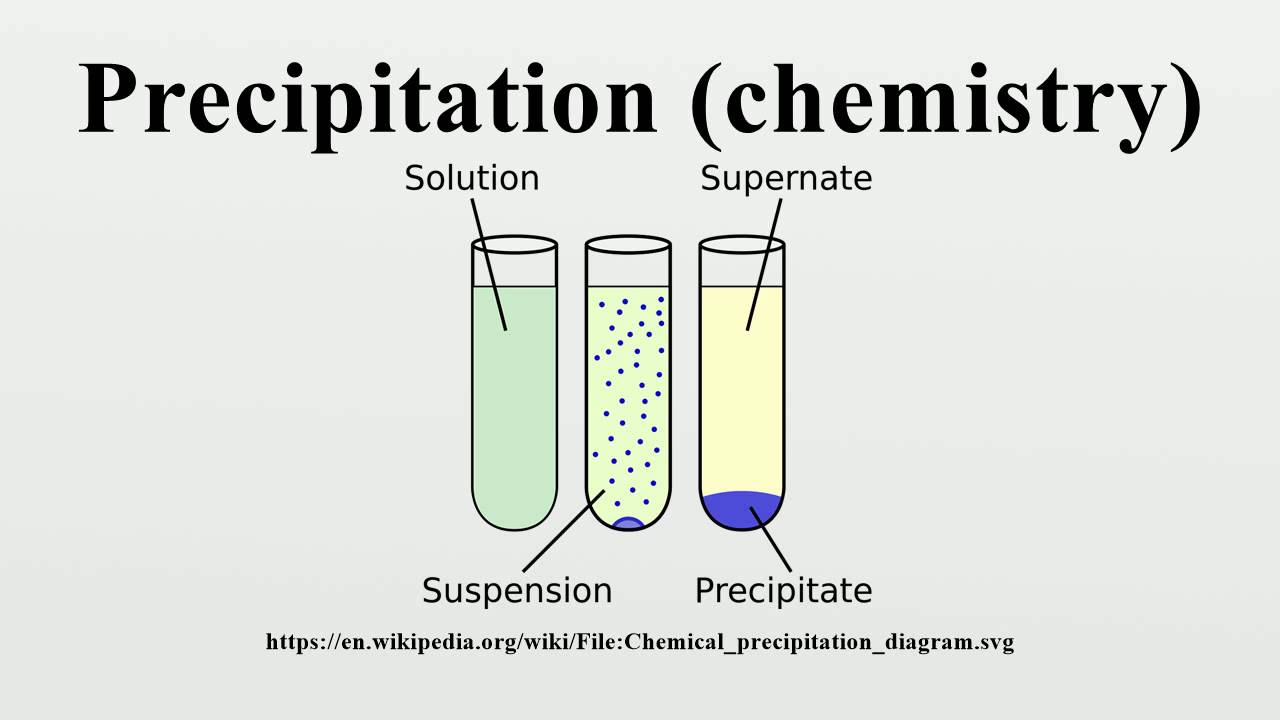
Chemical equations are a very important tool that can help us in understanding the chemical reactions between two or more elements or compounds. During a Chemical reaction, chemical changes occur that lead to the formation of new compounds under some specific conditions.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
